Applying the Hierarchy of Controls to the Seven Aspects of Surface Selection™
A copy of this article may be found in the October 2019 issue of Healthcare Hygiene Magazine.
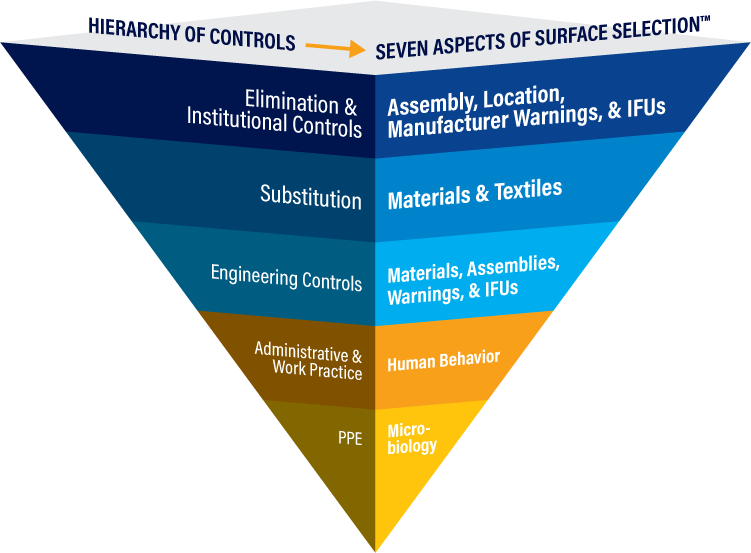
The Healthcare Surfaces Institute has recently adopted a combination of the Hierarchy of Controls and the Seven Aspects of Surface Selection™ as a quick guide for patient and healthcare worker safety as it relates to surface selection.
Healthcare surfaces have long been established as fomites – objects or materials to carry microorganisms that can cause infection and illness. Research has shown that microbes can live on “clean and disinfected” surfaces for days, weeks, and even months. How is this possible when surfaces in healthcare settings are regularly cleaned and disinfected?
Surfaces are complicated. Discussion around the impact surfaces have in infection prevention and control creates confusion and misunderstanding. Some believe environmental or “high touch” surfaces like sinks, counters, bedrails, door handles, light switches and patient equipment are the main contributing factor for the acquisition and spread of pathogens, but these surfaces only make up about 25% of surfaces in patient and procedure rooms.
Limiting focus on these surfaces and/or these thought process leaves about 75% of the surfaces that exist in the healthcare environment unaddressed. These include upholstery on furniture, walls, floors, linen, fixtures, and more. They also include surfaces made up of multiple surface materials that interact with patient and personnel including medical devices, surgical equipment, and instruments. In addition, soft surfaces such as scrubs, hospital gowns, and bedding always move between various areas of the hospital and can serve as fomites.
Seven Aspects of Surface Selection™
In order to reduce the role that healthcare surfaces play in the acquisition and transmission of pathogens, the “Seven Aspects of Surfaces Selection™” must be evaluated to address this issue proactively. Considerations include:
- Surface Materials and Textiles – There are many different surface materials and textiles used within healthcare facilities both in the built environment and products used during patient care.
- Assemblies – Multiple surface materials on any one product must be evaluated individually and as an assembly for use and cleanability. Examples; beds and other medical devices, furniture…
- Location – Location of the surfaces–high contamination and high turnover areas require durable surfaces that can withstand frequent disinfection. Example; OR, ED.
- Cleaning, Disinfection/Sterilization – Infection prevention protocol, process and products that will be used must be considered and an evaluation completed before products and surfaces are purchased and put into service.
- Human Behavior – Humans continually interact with surfaces and move throughout the healthcare facility. While hand hygiene is critical, clothing and other products also move throughout the facility. Many questions exist around human behavior and transmission of microbes that cause deadly infections.
- Microbiology – There are surface materials that support the proliferation of microbes despite routine cleaning. Manufacturers lack standardized testing for microbes that allow purchasers to compare products equitably.
- Manufacturer Warnings and Instructions for Use (IFUs) – Disinfectant compatibility testing for surfaces is rarely if ever done. It is concerning when manufacturers list all disinfectants as compatible without testing data and validation that they are in fact compatible creating risk of damage and the potential for a voided warranty for healthcare facilities. It is also important that healthcare professionals request manufacturing warnings. In this document, you will find warnings that include particular chemicals used in disinfectants and detergents.
Note: Many products have multiple surface materials that cannot all be cleaned, disinfected and sterilized the same way. When they are damage occurs creating reservoirs for microbes to harbor out of the reach of biocides.
By incorporating the Seven Aspects of Surface Selection into the Hierarchy of Controls, an innovative method begins to emerge for addressing surfaces more comprehensively. The process more closely integrates infection prevention and occupational health and allows healthcare institutions, manufacturers, and experts the ability to address controls for surfaces in a more methodical process with the focus first on making the biggest impact.
However, currently, there are no consistent surface testing standards or requirements. This can leave those accountable and responsible for infection prevention and patient care at risk. This is due in part to a general lack of understanding and knowledge about the impact surface materials and composition can have on the ability to successfully clean, disinfect, and/or sterilize them.
Hierarchy of Controls
Controlling exposures to occupational hazards is the fundamental method of protecting workers. While this concept is typically used in occupational health and industrial hygiene disciplines, we can use the hierarchy to apply the same concepts to protect patients and their caregivers from healthcare surface-derived hazards as well. Traditionally, a hierarchy of controls has been used as a means of determining how to implement feasible and effective control solutions in a manner that provides first, the greatest level of protection.
The idea behind this hierarchy is that the control methods at the top of the graphic (below) are more effective and protective than those at the bottom. Following this hierarchy leads to the implementation of inherently safer systems, where the risk of illness is substantially reduced.
The traditional occupational health hierarchy was formally created in the 1960s for use in nonhealthcare industries like manufacturing, mining, and construction. It is used as the basis of federal and state occupational safety and health regulations, including the Occupational Safety and Health Administration (OSHA). For example, the OSHA Bloodborne Pathogens Standard has resulted in unnecessary needles being eliminated wherever possible (e.g. needleless IV catheter connections), reducing the risk of needlesticks. If a needle cannot be eliminated, can it be substituted for something less hazardous? This might mean using blunt fill needles to draw up medications from multi-dose vials.
If a needle cannot be eliminated or substituted out, can it be engineered to be safer? Disposable hypodermic needles with sharps injury prevention features (e.g., sheathing or retracting needles) are a perfect example of engineering controls.
If a needle cannot be engineered to be safer, are there work practices or administrative controls that can be put into place to reduce sharps injuries? This includes safe disposal practices and careful attention to surgical team safety when using no-hands or neutral zones for surgical instruments.
If, finally, none of these controls can be put into place or there is still an exposure risk after the higher controls have been implemented, does personal protective equipment (PPE) need to be used? Does a single or double pair of gloves need to be worn? Is there risk of a blood splatter, necessitating a gown and/or eye protection be worn to protect someone at the patient bedside or performing a diagnostic test on a patient sample in a clinical lab?
PPE is the lowest and least effective control on the hierarchy for many reasons.
- It must be available to the healthcare worker when and where it is needed. It must be immediately accessible to them.
- The worker must make the decision to put it on.
- The worker must be properly educated about risk, so they are more likely to use it.
- The worker must be educated about how to properly put it on (don) and take it off (doff) and to safely dispose of PPE so as not to contaminate themselves or inadvertently expose anyone downstream (e.g., environmental services and waste haulers).
- The PPE must work. It cannot have gaps or tears or an undesirable level or permeability.
- Multiple pieces of PPE must be compatible. If a procedure can result in a risk of a blood or body fluid splash or splatter (mucocutaneous), can eye protection be worn in an addition to a respirator? Or is the isolation or chemo gown long enough to cover the wrists in addition to glove use?
- Cost and disposal are concerns. Since most PPE is disposable, providing PPE everywhere it is needed can be costly and if it is not properly disposed of, it can pose a risk to workers downstream. And since PPE is disposable, are there additional occupational or environmental concerns when PPE materials (e.g., latex, nitrile, vinyl) are made or when biohazard or red bag waste is incinerated?
In infection prevention and control and environmental safety and health, professionals have focused heavily on PPE use as required by contact, isolation, and/or transmission-based precautions. Why do we focus on PPE for infection prevention and patient safety, often as a first-line defense and not in occupational health and safety?
Why do healthcare institutions focus on work practices like properly cleaning and disinfecting environmental surfaces and cleaning and sterilizing surgical instruments and manufacturers are not held accountable and responsible for ensuring there materials and products meet the demanding requirements and needs of healthcare facilities and professionals.
There is a great deal that the infection prevention and environmental services community can learn from their occupational health and safety partners, including ways to integrate the hierarchy of controls into their processes, policies, and practices. Neither infection prevention nor occupational health have more resources than they need to reduce risks for the patients and providers they work to protect. The most effective programs are those that partner “across the aisle” and share not only resources, but expertise and experience.
(REF: https://www.cdc.gov/niosh/topics/hierarchy/default.html)
The Hierarchy of Controls:
-
Elimination: Physically remove the hazard; Institutional Controls: Culture of safety
-
Substitution: Replace the hazard
-
Engineering Controls: Isolate people from the hazard
-
Administrative Controls: Change the way people work and interactive with their environment
-
PPE: Protect the worker and patient with barrier controls
Integrating the application of the Hierarchy of Controls to the Seven Aspects of Surface Selection looks like this:

It is believed that to reduce the risk of healthcare associated infections (HAIs) and occupationally associated infections (OAIs) one must clean, disinfect, and sterilize surfaces, devices, instruments, textiles, and patient care items better. This certainly is one aspect of the solution. Unfortunately, it will not adequately address the problem.
When all aspects are included in the Hierarchy of Controls, a proactive strategy becomes clearer and more distinct. Patient and healthcare worker risks are minimized, and sustainable solutions and relationships can be realized.
Linda Lybert is president of Healthcare Surface Consulting and the founder/executive director of the Healthcare Surfaces Institute.
Amber Hogan Mitchell, DrPH, MPH, CPH, is president and executive director of the International Safety Center and a member of the board of the Healthcare Surfaces Institute.
Glenda Schuh, RN, BSN, CIC, and Caroline Etland, PhD, MSN, reviewed this article. Schuh is a consultant in infection prevention and occupational health. Etland is currently an associate professor at the University of San Diego Hahn School of Nursing, and a clinical nurse specialist at Sharp HealthCare. They both serve as board members of the Healthcare Surfaces Institute.

